Clinical Breast Cancer Trials Underway At Northwestern, Promising Drug To Treat Early Stages
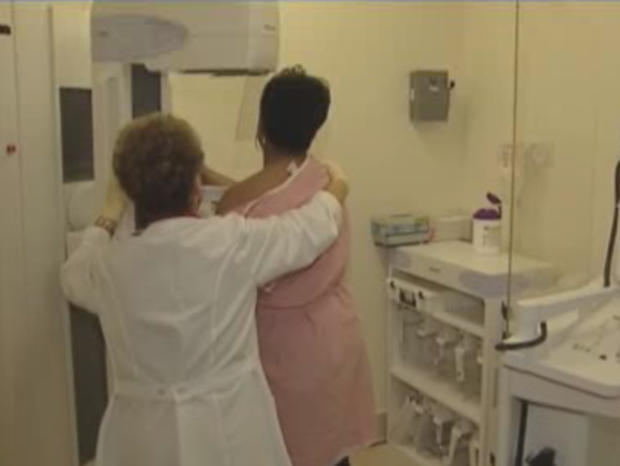
CHICAGO (CBS) -- Clinical trials are underway to test a medication that is already approved for menopausal symptoms but it also may prevent and better treat early stages of breast cancer.
"I'm working on breast cancer prevention and I recently opened this new clinical trail where I am studying a medication that is already approved for menopausal symptoms, so it already helps women with hot flashes, sleeping and osteoporosis, so it's already readily available," said Dr. Swati Kulkarni, Surgical Oncologist, Northwestern. "But one of the things we've learned about this medication is that it also may prevent breast cancer."
Kulkarni said if the medication reverses or slows the changes in the development of breast cancer, it could mean women could avoid surgery and radiation and most importantly prevent later stage breast cancer.
"This is exciting because we have a medication that makes you feel better, but it may prevent breast cancer."
Kalkarni is studying this medication in women who have noninvasive cancer, DCIS or ductal carcinoma in situ.
"What we're doing is that when women are diagnosed we ask them to participate in this study. They'll take the medication for three to five weeks before they have surgery. They'll be randomly put in the treatment group or the placebo group. We're going to enroll 160 women across the country and we're going to compare changes in their breast cancer before they take the medication and after they take the medication," she said.
Researchers will then analyze a small piece of their breast tissue and try to determine if this medication reverses or slows the changes in the breast that are associated with the development of invasive breast cancer.
"The great potential of it is if we find that this medication reverses or slows these changes, we may have an alternative in the future to our current treatment for noninvasive cancer with this surgery and sometimes radiation," she said.
DCIS affects about 20 percent of breast cancer patients.
"It's pretty common. It usually shows up on screening mammogram. It's considered stage zero breast cancer. They are abnormal cells in the milk ducts for the breasts. When we worry about cancer, we worry about cancer escaping from the breast and spreading to other parts of the body. DCIS usually don't have the potential to spread. It's really a precancerous lesion. About 30 percent of women can turn into an invasive breast cancer," Kalkarni said.
The trials opened in January and the hospital has been recruiting patients since January.
"We have good data from animal studies and shows that this is very promising." she said.
Northwestern is the lead site for these clinical trials. The hospital is looking for postmenopausal women willing to take part in the study.
Anyone with questions about cancer clinical trials at Northwestern Memorial Hospital should please call 312.695.1102 to talk with our clinical trial nurse specialist.